Overview
In 2020, we have successfully obtained the GSP Certificate,owned a professional team with over 40 employees, and a pharmaceutical storage warehouse that is about 2000㎡in size. The warehouse is equipped with normal temperature area and cooling area, and automatic temperature humidity sensor that monitors the warehouse constantly.
With the GSP certificate, our company is able to import API as well as pharmaceutical preparations and distribute them to qualified agencies&hospitals in China. So far, we have built close partnership with many large scale pharmaceutical factories in Korea, Japan, India, etc.
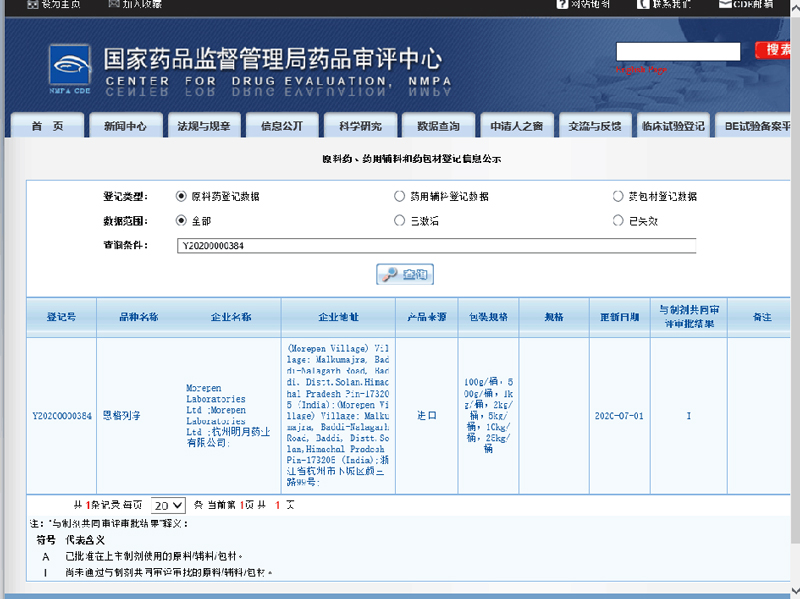
In the meantime, we are also registering pharmaceutical products with SFDA. The most recent API we registered is Empagliflozin, and there are more to be expected in the near future.